Unit 2 - Chemical Reactions & Radioactivity

Chemical reactions occur when matter undergoes changes. Usually something in the environment has changed to cause this reaction to occur. Something like temperature of pressure change, matter coming in contact with other matter or an electrical spark could ignite a chemical reaction.
Can you think of any chemical reactions you have seen in your life?
Most people have seen what happens when you mix baking soda and vinegar together. This is a chemical reaction.
Chapter 4: Atomic theory explains the formation of compounds
PLO's - differentiate between atoms, ions, and molecules using knowledge of their structure and components
Section 4.1 - Atomic Theory & Bonding
Section 4.2 - Names & Formulas of Compounds
Section 4.3 - Chemical Equations
PLO's - differentiate between atoms, ions, and molecules using knowledge of their structure and components
Section 4.1 - Atomic Theory & Bonding
Section 4.2 - Names & Formulas of Compounds
Section 4.3 - Chemical Equations
Atomic Theory and Bonding
Chemistry is the study of changes in matter. What is matter?
Matter is anything that has mass and takes up space. So pretty much everything that you can see is matter.
There is one thing that fills up a room, but has no mass and is not matter. Can you think of what it is?
What is matter made of? The particle theory of matter says that all matter is made of small particles and that these particles are in constant motion.
These particles can be molecules or atoms. Molecules are larger particles that are made up of different types of atoms bonded together.
Atoms are the smallest unit of matter. Matter cannot be broken down smaller than an atom and still have the properties that the matter was known for.
Atomic Theory
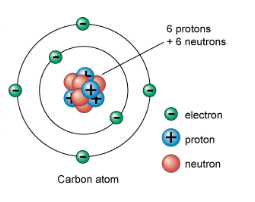
Atoms are composed of small particles: the proton, the electron and the neutron.
Protons: subatomic particles that have a positive charge (+) and are located in the nucleus.
Neutrons: subatomic particles that have a neutral charge (0) and are located in the nucleus.
Electrons: are subatomic particles that have a negative charge (-) and are found orbiting the nucleus.
Chemistry is the study of changes in matter. What is matter?
Matter is anything that has mass and takes up space. So pretty much everything that you can see is matter.
There is one thing that fills up a room, but has no mass and is not matter. Can you think of what it is?
What is matter made of? The particle theory of matter says that all matter is made of small particles and that these particles are in constant motion.
These particles can be molecules or atoms. Molecules are larger particles that are made up of different types of atoms bonded together.
Atoms are the smallest unit of matter. Matter cannot be broken down smaller than an atom and still have the properties that the matter was known for.
Atomic Theory
Atoms are composed of small particles: the proton, the electron and the neutron.
Protons: subatomic particles that have a positive charge (+) and are located in the nucleus.
Neutrons: subatomic particles that have a neutral charge (0) and are located in the nucleus.
Electrons: are subatomic particles that have a negative charge (-) and are found orbiting the nucleus.
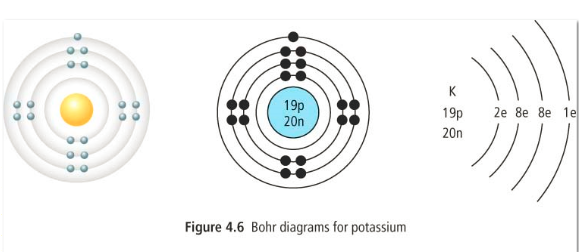
The nucleus of an atom contains the protons and the neutrons. These two particles are what give the atom its mass. The nucleus is always positively charged as the protons are positive and the neutrons are neutral. The number of protons in an atom gives the atom its atomic number. What is the atomic number of the carbon atom above?
In a neutral atom, the number of electrons equals the number of protons. This means that the atom as an overall charge of zero.
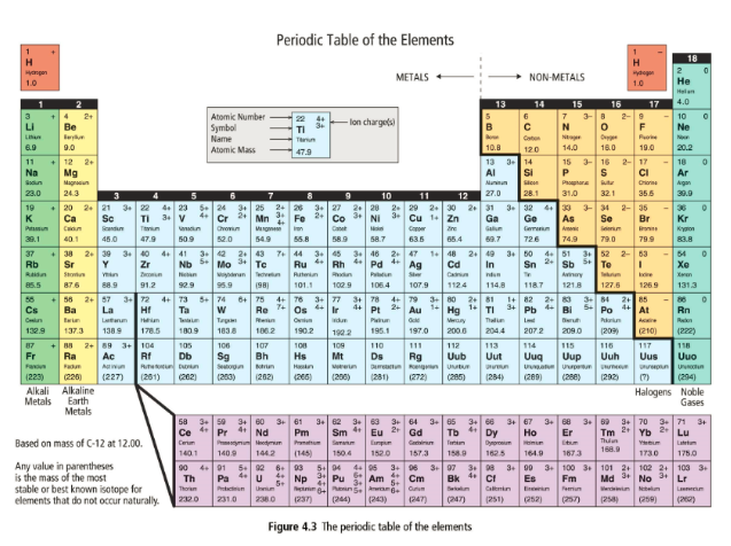
Organization of the Periodic Table of the Elements
One single type of atom is called an element. All of the elements known to exists are organized according to their properties on the periodic table. You should print a copy of this table from the data pages as you will be referring to it a great deal in this unit.
The atoms are organized by increasing atomic number. There are rows (periods) and columns (groups or family). The smallest atom is hydrogen, in the top left corner. It has an atomic number of 1. The largest atom is ununoctium, in the bottom right corner and it has an atomic number of 118.
Atoms in the same family have similar properties. For example, all the atoms in group 18 are noble gases. This means that they are all gases at room temperature and they are non-reactive.
Metals and non-metal are separated by the thick black zig-zag line. Non-metals are on the right and metals are on the left.
In a neutral atom, the number of electrons equals the number of protons. This means that the atom as an overall charge of zero.
Organization of the Periodic Table of the Elements
One single type of atom is called an element. All of the elements known to exists are organized according to their properties on the periodic table. You should print a copy of this table from the data pages as you will be referring to it a great deal in this unit.
The atoms are organized by increasing atomic number. There are rows (periods) and columns (groups or family). The smallest atom is hydrogen, in the top left corner. It has an atomic number of 1. The largest atom is ununoctium, in the bottom right corner and it has an atomic number of 118.
Atoms in the same family have similar properties. For example, all the atoms in group 18 are noble gases. This means that they are all gases at room temperature and they are non-reactive.
Metals and non-metal are separated by the thick black zig-zag line. Non-metals are on the right and metals are on the left.
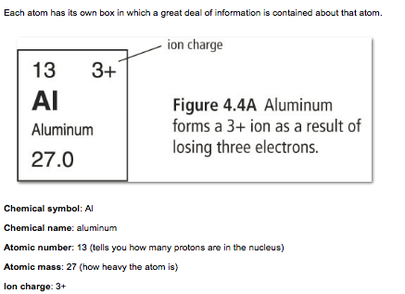
Electrons an be gained or lost by an atom. When this happens, an ion is formed. If an atom loses electrons, then the protons are greater in number than the electrons and the atom will have a positive charge (cation). If the atom gains electrons then the number of electrons will be greater than the number of protons and the atom will have a negative charge (anion).
Bohr Diagrams
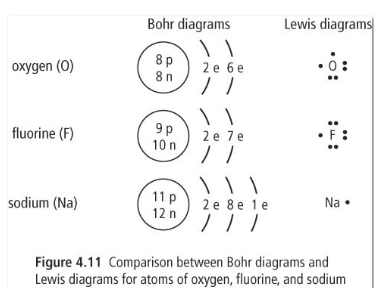
These are diagrams of atoms that show all electrons and their organization into energy levels.
Steps for drawing a Bohr diagram:
1. Write the number of protons in the center with a circle around it. This is the nucleus.
2. Draw another circle around the nucleus. This is the first layer of electrons. This layer can only hold 2 electrons and they are drawn in the north position of the circle as a pair.
3. We need to draw another circle around the first and this layer can hold a max of 8 electrons. We draw them one at a time in the north then east, then south, then west positions. If we have more electrons then we start the circle over again, pairing the electrons up.
4. Continue this process until all the electrons in the atom are organized.
Draw Bohr diagrams for lithium, carbon and magnesium atoms as practice.
The last shell of electrons is called the valence shell. It contains the valence electrons. How many valence electrons are present in the Bohr diagrams you completed above?
The number of valence electrons will tell you how the atom will form its ion.
Atoms are stable and happy when they have a full eight electrons in their valence shells. This is termed a stable octet.
Atoms will either give away or take electrons in order to achieve their stable octet.
We can draw Bohr diagrams of ions by starting with the diagrams of the atoms, then either filling in the valence shell if there is 5 or more valence electrons already present, or erasing the valence electrons if there is less than 4 already present.
Once that is done, we draw square brackets around the diagram and write the ion charge outside the top right corner of the bracket.
Draw Bohr diagrams for lithium, and magnesium ions as practice
Atomic Bonding
Atoms can join together to form bonds. Two types of bonds are possible and we can draw Bohr diagrams for each type.
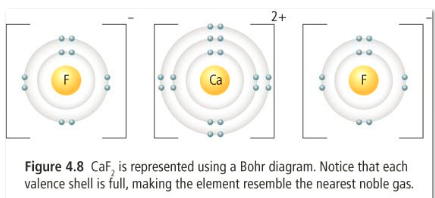
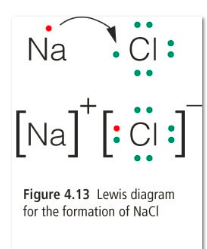
1) Ionic bonds: ions that have opposite charges are attracted to each other and held together by this attraction. Ions form ionic bods in such a way that the over all charge is zero. This type of bond occurs between a metal and a non-metal.
1. Write the number of protons in the center with a circle around it. This is the nucleus.
2. Draw another circle around the nucleus. This is the first layer of electrons. This layer can only hold 2 electrons and they are drawn in the north position of the circle as a pair.
3. We need to draw another circle around the first and this layer can hold a max of 8 electrons. We draw them one at a time in the north then east, then south, then west positions. If we have more electrons then we start the circle over again, pairing the electrons up.
4. Continue this process until all the electrons in the atom are organized.
Draw Bohr diagrams for lithium, carbon and magnesium atoms as practice.
The last shell of electrons is called the valence shell. It contains the valence electrons. How many valence electrons are present in the Bohr diagrams you completed above?
The number of valence electrons will tell you how the atom will form its ion.
Atoms are stable and happy when they have a full eight electrons in their valence shells. This is termed a stable octet.
Atoms will either give away or take electrons in order to achieve their stable octet.
We can draw Bohr diagrams of ions by starting with the diagrams of the atoms, then either filling in the valence shell if there is 5 or more valence electrons already present, or erasing the valence electrons if there is less than 4 already present.
Once that is done, we draw square brackets around the diagram and write the ion charge outside the top right corner of the bracket.
Draw Bohr diagrams for lithium, and magnesium ions as practice
Atomic Bonding
Atoms can join together to form bonds. Two types of bonds are possible and we can draw Bohr diagrams for each type.
1) Ionic bonds: ions that have opposite charges are attracted to each other and held together by this attraction. Ions form ionic bods in such a way that the over all charge is zero. This type of bond occurs between a metal and a non-metal.
In this example, two fluorine ions are needed to bond with1 calcium ion in order to make the overall charge zero. Like charges repel each other, so the fluorine ions are as far apart as possible.
Draw a Bohr diagram for NaCl and MgCl2 as practice
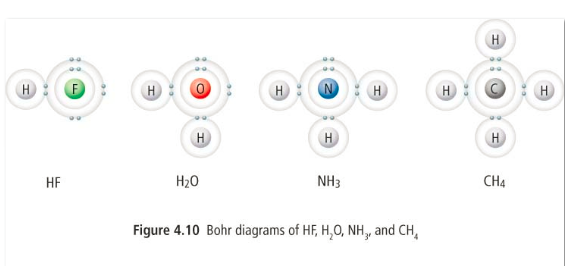
2) Covalent bonding: atoms can share electrons in order to fill their valence shells and have a stable octet. This type of bond is stronger than an ionic bond and occurs between two non-metals.
When we draw Bohr diagrams of covalent molecules, we draw them with their valence shells overlapped and the lone electrons from each atom paired up.
Draw a Bohr diagram for NaCl and MgCl2 as practice
2) Covalent bonding: atoms can share electrons in order to fill their valence shells and have a stable octet. This type of bond is stronger than an ionic bond and occurs between two non-metals.
When we draw Bohr diagrams of covalent molecules, we draw them with their valence shells overlapped and the lone electrons from each atom paired up.
Draw Bohr diagrams for the following covalent molecules: HCl, H2S
Lewis Diagrams
Another method of drawing atoms is called the Lewis diagram. These are much simpler and only involve writing the atomic symbol and then arranging the valence electrons around the symbol in the same method as the Bohr diagrams used.
Draw Lewis Diagrams for lithium, boron and sulfur atoms as practice
Ions in Lewis Diagrams
Ions are drawn with either the valence shell empty, if the atom lost its valence electrons or with the valence shell full, it the atom gained electrons in order to have a stable octet.
Ionic bonds are shown in the same way as Bohr diagrams. The ions are drawn side b y side in brackets with the ion charge outside the top right corner of the bracket.
Ions in Lewis Diagrams
Ions are drawn with either the valence shell empty, if the atom lost its valence electrons or with the valence shell full, it the atom gained electrons in order to have a stable octet.
Ionic bonds are shown in the same way as Bohr diagrams. The ions are drawn side b y side in brackets with the ion charge outside the top right corner of the bracket.
Draw lewis diagrams of LiCl, NaF and MgCl2
Covalent bonds are shown with the lone electrons pairing up, then that pair is replaced in the diagram with a straight line representing the bond.
Covalent bonds are shown with the lone electrons pairing up, then that pair is replaced in the diagram with a straight line representing the bond.
Draw lewis diagrams for H2O, CH4, H2S
| bohr__lewis_worksheet.pdf | |
| File Size: | 34 kb |
| File Type: | |
QUIZ
Names and Formulas of Compounds
Practice Naming & Writing
When atoms combine to form ionic compounds and covalent molecules, we use a set of rules to determine what the name or the formula of the compound or molecule is.
Naming Ionic Compounds
Ionic compounds contain a metal and a non-metal. Here are the steps to naming an ionic compound:
1. Name the metal first. It is always the first atomic symbol in the formula
2. Name the non-metal second, but change the last syllable to "-ide"
For example, NaCl: sodium is the metal, so it is named first. Chlorine is the non-metal, so we change the name chlorine to chloride
3. Put the two names together: Sodium Chloride
KBr would be called potassium bromide
Complete the Practice Problems on the bottom of page 187
Writing the Formulas of Ionic Compounds
If we are given the name of an ionic compound we should be able to determine the chemical formula if we follow the steps below.
1. Identify each ion and its charge.
2. Determine how many ions each are needed to achieve an overall charge of zero.
3. Write the metal(cation) ion symbol first followed by the number needed in subscript, then write the non-metal (anion) symbol next followed by the number needed in subscript.
*note: if the number needed is 1, no subscript is required.
example: aluminum fluoride.
1. Al 3+ F-
2. So in order to achieve a net zero charge, I will need 1 Al and 3 F.
3. AlF3
example: magnesium nitride
1. Mg 2+ N 3-
2. So in order to achieve a net zero charge I would need 3 Mg and 2 N.
3. Mg2N3
Do the practice problems on the bottom of page188
Writing the Formula of an Ionic Compound Containing a Multivalent Metal
Multivalent metals are metals that can have more than one ion charge. If you look at the periodic table, you should be able to find metals that have more than one number in the upper right corner of their box.
The first metals in the families 4 through 11 are all multivalent. Manganese has 3 possible charges, 2+, 3+, 4+
When the name of a compound containing a multivalent metal is given, there must be a roman numeral beside the name of the metal to indicate which charge is being used.
For example: Nickel (II) Oxide, the roman numeral II tells us that nickel is in its 2+ state.
Now we can write the formula as we did before .
NiO
Write the formulas for Cobalt (III) Nitride, Chromium (III) Oxide and Copper (I) Phosphide
Writing the Name of an Ionic Compound Containing a Multivalent Metal
If we are given the formula for an ionic compound containing a multivalent metal, we must deduce the charge on the metal by looking at the ratio of atoms and knowing that compounds always combine to have a net zero charge.
Lets look at Fe2O3
- we know that Fe can have a 2+ or a 3+ charge
- we know that O can only have a 2- charge.
- we know that there are 6 negative charges in Fe2O3 because there are 3 oxygen atoms, each with a 2- charge.
- there are only 2 Fe atoms in the compound that must combine to equal 6+ charges, so each of them must be 3+.
Therefore, Fe2O3 can be named as Iron (III) Oxide
Write the names for TiO2, CuF, Au2S3
Complete the Practice Problems on the bottom of page 190 and 191
Writing the Formula of a Compound Containing a Polyatomic Ion
Look at table 4.11 on page 192. These are ions that have more than one atom in them. We call them polyatomic ions.
If a molecule has more than 2 elements in it, chances are it has one of these polyatomic ions in it.
If you cannot find one of the names on the periodic table, then you probably have a polyatomic ion.
NaOH: this molecule has 3 elements in it.
OH- is the hydroxide ion, so we need to use this name in our chemical name.
Sodium Hydroxide
Mg3(PO4)2
This molecule also has 3 elements in it, meaning we should look at our polyatomic ion chart.
We find that PO4 is the phosphate ion.
So the name is magnesium phosphate.
Complete the Practice Problems on page 193.
Names and Formulas of Covalent Compounds
First we need to recognize a molecule as being covalent before we set into motion the steps outlined below. There are no metals in covalent compounds. So all elements present must be non-metals.
When we write the name of a covalent compound, we use prefixes to show the number of atoms in the molecule. The prefixes used are listed below:
Mono - 1
Di- 2
Tri - 3
Tetra - 4
Penta - 5
Hexa - 6
Hepta - 7
Octa - 8
Nona - 9
Deca - 10
Lets look at a couple examples:
CO: We can clearly see that there is one atom of carbon present and one atom of oxygen present. If the first atom listed only has one atom, we do not use the prefix mono-. Only with the second atom do we use mono.
Carbon Monoxide is the name for CO.
CO2: carbon dioxide
P4O10: tetraphosphorous decaoxide
If we are given the names, we can use the prefixes to indicate which subscripts to write.
Carbon tetrachloride: CCl4
dinitrogen tetrasulfide: N2S4
Practice Naming & Writing
When atoms combine to form ionic compounds and covalent molecules, we use a set of rules to determine what the name or the formula of the compound or molecule is.
Naming Ionic Compounds
Ionic compounds contain a metal and a non-metal. Here are the steps to naming an ionic compound:
1. Name the metal first. It is always the first atomic symbol in the formula
2. Name the non-metal second, but change the last syllable to "-ide"
For example, NaCl: sodium is the metal, so it is named first. Chlorine is the non-metal, so we change the name chlorine to chloride
3. Put the two names together: Sodium Chloride
KBr would be called potassium bromide
Complete the Practice Problems on the bottom of page 187
Writing the Formulas of Ionic Compounds
If we are given the name of an ionic compound we should be able to determine the chemical formula if we follow the steps below.
1. Identify each ion and its charge.
2. Determine how many ions each are needed to achieve an overall charge of zero.
3. Write the metal(cation) ion symbol first followed by the number needed in subscript, then write the non-metal (anion) symbol next followed by the number needed in subscript.
*note: if the number needed is 1, no subscript is required.
example: aluminum fluoride.
1. Al 3+ F-
2. So in order to achieve a net zero charge, I will need 1 Al and 3 F.
3. AlF3
example: magnesium nitride
1. Mg 2+ N 3-
2. So in order to achieve a net zero charge I would need 3 Mg and 2 N.
3. Mg2N3
Do the practice problems on the bottom of page188
Writing the Formula of an Ionic Compound Containing a Multivalent Metal
Multivalent metals are metals that can have more than one ion charge. If you look at the periodic table, you should be able to find metals that have more than one number in the upper right corner of their box.
The first metals in the families 4 through 11 are all multivalent. Manganese has 3 possible charges, 2+, 3+, 4+
When the name of a compound containing a multivalent metal is given, there must be a roman numeral beside the name of the metal to indicate which charge is being used.
For example: Nickel (II) Oxide, the roman numeral II tells us that nickel is in its 2+ state.
Now we can write the formula as we did before .
NiO
Write the formulas for Cobalt (III) Nitride, Chromium (III) Oxide and Copper (I) Phosphide
Writing the Name of an Ionic Compound Containing a Multivalent Metal
If we are given the formula for an ionic compound containing a multivalent metal, we must deduce the charge on the metal by looking at the ratio of atoms and knowing that compounds always combine to have a net zero charge.
Lets look at Fe2O3
- we know that Fe can have a 2+ or a 3+ charge
- we know that O can only have a 2- charge.
- we know that there are 6 negative charges in Fe2O3 because there are 3 oxygen atoms, each with a 2- charge.
- there are only 2 Fe atoms in the compound that must combine to equal 6+ charges, so each of them must be 3+.
Therefore, Fe2O3 can be named as Iron (III) Oxide
Write the names for TiO2, CuF, Au2S3
Complete the Practice Problems on the bottom of page 190 and 191
Writing the Formula of a Compound Containing a Polyatomic Ion
Look at table 4.11 on page 192. These are ions that have more than one atom in them. We call them polyatomic ions.
If a molecule has more than 2 elements in it, chances are it has one of these polyatomic ions in it.
If you cannot find one of the names on the periodic table, then you probably have a polyatomic ion.
NaOH: this molecule has 3 elements in it.
OH- is the hydroxide ion, so we need to use this name in our chemical name.
Sodium Hydroxide
Mg3(PO4)2
This molecule also has 3 elements in it, meaning we should look at our polyatomic ion chart.
We find that PO4 is the phosphate ion.
So the name is magnesium phosphate.
Complete the Practice Problems on page 193.
Names and Formulas of Covalent Compounds
First we need to recognize a molecule as being covalent before we set into motion the steps outlined below. There are no metals in covalent compounds. So all elements present must be non-metals.
When we write the name of a covalent compound, we use prefixes to show the number of atoms in the molecule. The prefixes used are listed below:
Mono - 1
Di- 2
Tri - 3
Tetra - 4
Penta - 5
Hexa - 6
Hepta - 7
Octa - 8
Nona - 9
Deca - 10
Lets look at a couple examples:
CO: We can clearly see that there is one atom of carbon present and one atom of oxygen present. If the first atom listed only has one atom, we do not use the prefix mono-. Only with the second atom do we use mono.
Carbon Monoxide is the name for CO.
CO2: carbon dioxide
P4O10: tetraphosphorous decaoxide
If we are given the names, we can use the prefixes to indicate which subscripts to write.
Carbon tetrachloride: CCl4
dinitrogen tetrasulfide: N2S4
|
|
|
| ||||||||||||||||||||||||
Chemical Equations
When two different types of matter combine and change, we call this a chemical reaction.
When two different types of matter combine and change, we call this a chemical reaction.

In this chemical reaction, we can see 2 clear liquids being mixed together and a yellow substance being formed. We know that a chemical reaction has occurred, because of the change we are seeing.
The substances being combined we call reactants. They are doing the reacting.
The substances being produced we call the products.
We can describe these chemical reactions using equations, kind of like math equation, but with chemistry.
Equations can have words: sodium bicarbonate + hydrogen acetate -----> carbon dioxide + sodium acetate
Equations can have symbols: NaHCO3(s) + CH3COOH(l) -----> CO2(g) + NaCH3COO(l) + H2O(l)
The substances being combined we call reactants. They are doing the reacting.
The substances being produced we call the products.
We can describe these chemical reactions using equations, kind of like math equation, but with chemistry.
Equations can have words: sodium bicarbonate + hydrogen acetate -----> carbon dioxide + sodium acetate
Equations can have symbols: NaHCO3(s) + CH3COOH(l) -----> CO2(g) + NaCH3COO(l) + H2O(l)
Chapter 5: Compounds are classified in different ways
PLO's - classify substances as acids, bases, or salts, based on their characteristics, name, and formula; distinguish between organic and inorganic compounds
Section 5.1 - Acids and Bases
Section 5.2 - Salts, Organic Compounds
PLO's - classify substances as acids, bases, or salts, based on their characteristics, name, and formula; distinguish between organic and inorganic compounds
Section 5.1 - Acids and Bases
Section 5.2 - Salts, Organic Compounds
Salts
Salts are a group of chemicals that can be formed by reacting an acid with a base.
Salts are a very useful group of chemicals
The table salt we sprinkle on our foods is sodium chloride. NaCl.
NaCl is formed by the reaction of hydrochloric acid and sodium hydroxide.
HCl + NaOH -------> NaCl + HOH
This illustrates the pattern that we see whenever we react an acid with a base. The products are always a salt and water.
acid + base -------> salt + water
Salts are a group of chemicals that can be formed by reacting an acid with a base.
Salts are a very useful group of chemicals
The table salt we sprinkle on our foods is sodium chloride. NaCl.
NaCl is formed by the reaction of hydrochloric acid and sodium hydroxide.
HCl + NaOH -------> NaCl + HOH
This illustrates the pattern that we see whenever we react an acid with a base. The products are always a salt and water.
acid + base -------> salt + water
QUIZ
Organic Compounds
When you think of the word "organic" most people would associate life with the definition some how. Organic being derived from something that is or once was alive.
So it is with the next class of chemicals. Organic chemicals are chemicals that contain carbon and can be produced by living organisms.
Science has come a long way in the field of chemistry and now laboratories can produce these carbon compounds as well.
When you think of the word "organic" most people would associate life with the definition some how. Organic being derived from something that is or once was alive.
So it is with the next class of chemicals. Organic chemicals are chemicals that contain carbon and can be produced by living organisms.
Science has come a long way in the field of chemistry and now laboratories can produce these carbon compounds as well.
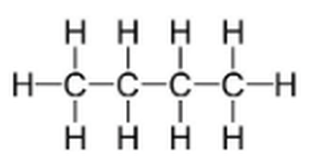
The molecule above is an organic molecule. It shows the Lewis diagram for the formula C4H10
We can recognize that the molecule above is organic because it contains carbon and hydrogen.
Very few molecules that contain carbon are not organic.
-If the carbon is part of a polyatomic ion, it is not an organic molecule.
-If the molecule is an oxide of carbon (carbon monoxide, CO, or carbon dioxide, CO2)
Hydrocarbons
These are a group of organic molecules that only contain the elements hydrogen and carbon.
All hydrocarbons are flammable and are often used as fuel. Households burn methane (natural gas). Butane and propane are sed as camp stove fuels and lighter fuels.
See table 5.8 for a list of the first 5 hydrocarbons
The molecule shown above is a hydrocarbon.
Alcohols: These are a group of organic molecules that contain only the elements carbon, hydrogen and oxygen. They are used as solvents, disinfectants and as antifreeze.
See table 5.9 for a list of some common alcohols.
QUIZ
We can recognize that the molecule above is organic because it contains carbon and hydrogen.
Very few molecules that contain carbon are not organic.
-If the carbon is part of a polyatomic ion, it is not an organic molecule.
-If the molecule is an oxide of carbon (carbon monoxide, CO, or carbon dioxide, CO2)
Hydrocarbons
These are a group of organic molecules that only contain the elements hydrogen and carbon.
All hydrocarbons are flammable and are often used as fuel. Households burn methane (natural gas). Butane and propane are sed as camp stove fuels and lighter fuels.
See table 5.8 for a list of the first 5 hydrocarbons
The molecule shown above is a hydrocarbon.
Alcohols: These are a group of organic molecules that contain only the elements carbon, hydrogen and oxygen. They are used as solvents, disinfectants and as antifreeze.
See table 5.9 for a list of some common alcohols.
QUIZ
Chapter 6: Chemical reactions occur in predictable ways
PLO's - analyze chemical reactions, including reference to conservation of mass and rate of reaction
Section 6.1 - Types of Chemical Reactions
Section 6.2 - Factors Affecting the Rate of Chemical Reactions
PLO's - analyze chemical reactions, including reference to conservation of mass and rate of reaction
Section 6.1 - Types of Chemical Reactions
Section 6.2 - Factors Affecting the Rate of Chemical Reactions
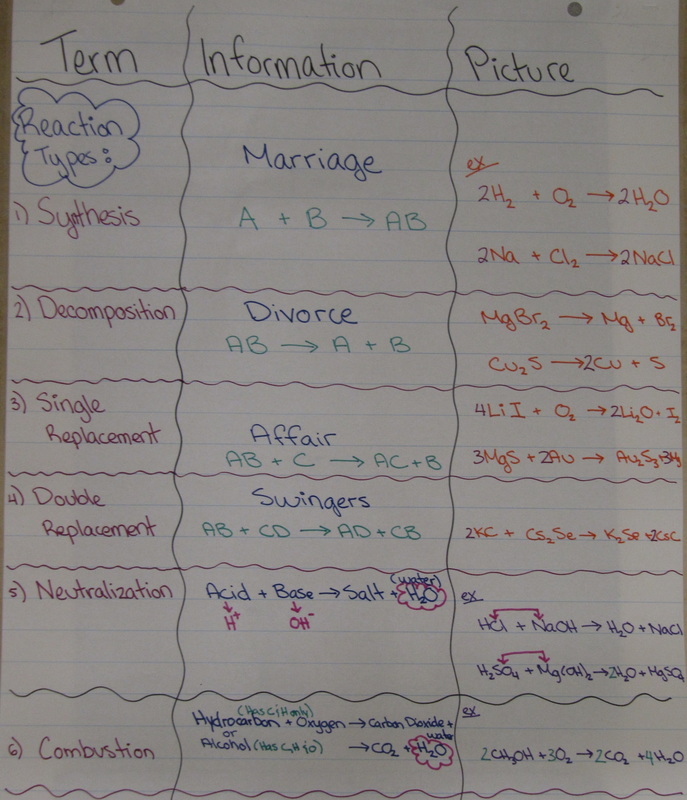
Types of Chemical Reactions
Anyone who has baked before knows that if you combine the same ingredients the same way you will get the same result every time.
The same is true in the chemistry lab. Chemical reactions proceed in a predicable way.
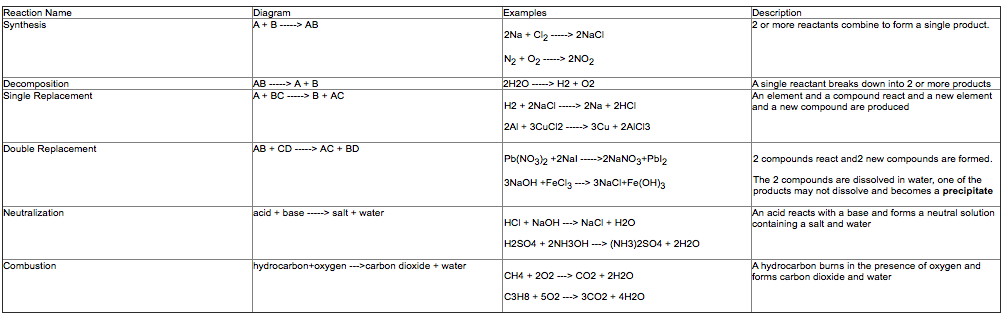
There are ways that chemists can organize chemical reactions in order to better predict the products. We can organize them into 6 types.
Anyone who has baked before knows that if you combine the same ingredients the same way you will get the same result every time.
The same is true in the chemistry lab. Chemical reactions proceed in a predicable way.
There are ways that chemists can organize chemical reactions in order to better predict the products. We can organize them into 6 types.
QUIZ
Factors Affecting the Rate of Chemical Reactions
When a chemical reaction happens and reactants form products, those products can be formed very quickly (like in an explosion), or very slowly (rust formation).
Fast reactions have a high rate of reaction
Slow reactions have a low rate of reaction
There are many things that determine how fast the products of a reaction will form. Such as:
Temperature
Concentration
Surface Area
Presence of a Catalyst
When a chemical reaction happens and reactants form products, those products can be formed very quickly (like in an explosion), or very slowly (rust formation).
Fast reactions have a high rate of reaction
Slow reactions have a low rate of reaction
There are many things that determine how fast the products of a reaction will form. Such as:
Temperature
Concentration
Surface Area
Presence of a Catalyst
Chapter 7: The atomic theory explains radioactivity
PLO's - explain radioactivity using modern atomic theory
Section 7.1 - Atomic Theory, Isotopes, and Radioactive Decay
Section 7.2 - Half-Life
Section 7.3 - Nuclear Reactions
PLO's - explain radioactivity using modern atomic theory
Section 7.1 - Atomic Theory, Isotopes, and Radioactive Decay
Section 7.2 - Half-Life
Section 7.3 - Nuclear Reactions
Unit 2 Review
This assignment contains questions from all sections of out unit in chemistry. Being able to complete all questions means that you are well prepared for the unit test.
In your text book, on pages 332 - 335 are a number of questions that test the skills you have learned in this section.
The following is a list of questions you need to be able to do in order to do well on the unit test.
2 - 4, 7 - 9, 11 - 14, 16, 17, 18, 20 - 27, 39, 43.
If you do not know how to do these questions, please ask for help. We will not have time to hand them in for marks before the test. So it will be up to you to ask your instructor if you have a question correct and are able to move on to the next one.
Good luck!
This assignment contains questions from all sections of out unit in chemistry. Being able to complete all questions means that you are well prepared for the unit test.
In your text book, on pages 332 - 335 are a number of questions that test the skills you have learned in this section.
The following is a list of questions you need to be able to do in order to do well on the unit test.
2 - 4, 7 - 9, 11 - 14, 16, 17, 18, 20 - 27, 39, 43.
If you do not know how to do these questions, please ask for help. We will not have time to hand them in for marks before the test. So it will be up to you to ask your instructor if you have a question correct and are able to move on to the next one.
Good luck!