|
| ||||||||||||
|
Part 5: DNA Replication - describe DNA replication and recombinant DNA.
Chapter 12: DNA and RNA Section 12.1 - DNA Section 12.2 - Chromosomes and DNA Replication |
Video: Section 12.1
Video: Section 12.2
|
Youtube - Through the Virtual Cell (6.48min)
|
Structure of DNADNA (deoxyribonucleic acid) is composed of polynucleotides (poly = many nucleotides). Each nucleotide is made up of three parts: a phosphate group, deoxyribose (a pentose sugar), and a base that contains nitrogen. The base may have a double ring structure (called purines) or a single ring structure (called a pyrimidine). The purine bases are adenine (A) and guanine (G); the pyrimidine bases are thymine (T) and cytosine (C). Adenine is always paired with thymine, and guanine is always bonded to cytosine. This is known as complementary base pairing.
Polynucleotides are joined to form long strands. Two of these strands, wound around each other and held together by hydrogen bonds, form the double helix shape of the DNA molecule. When unwound, DNA resembles a ladder. The phosphate groups and deoxyribose sugars form the sides, and weak hydrogen bonds between complementary base pairs complete the rungs of the ladder. Functions of DNADNA stores an organism's genetic information in the nuclei of cells. It also replicates (makes an exact copy of itself) when cells divide, so the nuclei of new cells contains the same DNA as the cells from which they originated. Finally, DNA provides a code or template for the particular sequencing of amino acids that ultimately bond together to form a protein.
|
Structure of RNARNA (ribonucleic acid), like DNA, is a polynucleotide. However, its structure is somewhat different from that of DNA. RNA is single-stranded, so it does not form a double helix. Instead of deoxyribose sugar, RNA contains ribose sugar. And while DNA does not leave the nucleus, RNA does move out of the nucleus to perform its roles in protein synthesis. Finally, in RNA, adenine pairs with uracil, a different base that replaces thymine.
Functions of RNAThere are three types of RNA and each is involved in protein synthesis. This topic will be explored in more depth in a future lesson. For now, it is important to know the basic functions of each type of RNA:
Messenger RNA (mRNA)
|
Comparing DNA & RNA Activity
How Does Replication Occur?
DNA Replication is the process of exactly copying one DNA double helix to produce two identical helices. The process is semi-conservative, meaning that each of the two strands from the original double helix provides a template for the formation of a complementary new strand. After replication, each new double helix will contain one original and one newly made strand.
Step 1: DNA unzips
Step 3: Replication is completed
Step 1: DNA unzips
- Recall that two DNA strands are held together by weak hydrogen bonding between base pairs—adenine is paired with thymine, cytosine with guanine). There are two hydrogen bonds, or H-bonds, between A and T and three between C and G. An enzyme, DNA helicase (enzyme names end in -ase), targets certain sections of DNA that contain a large proportion of A–T bonds. DNA helicase breaks the H-bonds, causing the DNA to unzip and unwind into two separate strands. This happens simultaneously in a number of locations.
- The action of DNA helicase results in two separated DNA strands with exposed bases that are no longer paired. Free-floating nucleotides in the nucleus pair up with the exposed bases on each strand (recall that adenine pairs with thymine, and cytosine pairs with guanine). These nucleotides are joined together by DNA polymerase to form a new sugar-phosphate backbone and create a new DNA strand.
Step 3: Replication is completed
- After the unattached nucleotides become attached to an unzipped DNA strand, DNA ligase seals any remaining breaks in the sugar-phosphate backbone. Two DNA double helices have then been created from one original DNA helix. Each new double helix contains one original and one newly made strand of DNA.
Part 6: Protein Synthesis - demonstrate an understanding of the process of protein synthesis, explain how mutations in DNA affect protein synthesis.
Chapter 12: DNA and RNA
Section 12.3 - RNA and Protein Synthesis
Section 12.4 - Mutations
Section 12.5 - Gene Regulation
Chapter 12: DNA and RNA
Section 12.3 - RNA and Protein Synthesis
Section 12.4 - Mutations
Section 12.5 - Gene Regulation
|
Videos: Section 12.4
|
|
Protein Synthesis
Part 1: Transcription
The structure of the DNA molecule is the same in every living organism. All of life, in its infinite variety, from slime mould to the extinct Stegosaurus, is constructed from the same basic information contained in each cell. How is it possible that a molecule consisting of only six parts can provide all the information needed to produce millions of species of life?
You have already learned about the structure of DNA and that it is made up of a long series of nucleotides. These nucleotides are made up of a deoxyribose sugar, a phosphate group, and a nitrogenous base (A, T, C, or G)—the six parts of the DNA molecule. The bases are paired (A with T and C with G) and joined by hydrogen bonds, and the sugars and phosphate groups form the backbone of DNA's three-dimensional double helix.
DNA codes for amino acids, which are the building blocks of proteins. Proteins, in turn, have a number of functions, including support, structure, movement, transport, communication, and immune defense. Protein-containing structures include hair, nails, hooves, horns, hormones, antibodies, blood proteins, and enzymes.
The structure of the DNA molecule is the same in every living organism. All of life, in its infinite variety, from slime mould to the extinct Stegosaurus, is constructed from the same basic information contained in each cell. How is it possible that a molecule consisting of only six parts can provide all the information needed to produce millions of species of life?
You have already learned about the structure of DNA and that it is made up of a long series of nucleotides. These nucleotides are made up of a deoxyribose sugar, a phosphate group, and a nitrogenous base (A, T, C, or G)—the six parts of the DNA molecule. The bases are paired (A with T and C with G) and joined by hydrogen bonds, and the sugars and phosphate groups form the backbone of DNA's three-dimensional double helix.
DNA codes for amino acids, which are the building blocks of proteins. Proteins, in turn, have a number of functions, including support, structure, movement, transport, communication, and immune defense. Protein-containing structures include hair, nails, hooves, horns, hormones, antibodies, blood proteins, and enzymes.
COPY CODE SNIPPET
In the next two lessons you will investigate protein synthesis, the complex process of creating a protein from a DNA sequence. Two major processes are involved:
In this lesson you will learn about the process of transcription, and begin to focus on how the information contained in DNA is processed to form a final product—a protein.
DNA is housed in the nucleus of a cell. In order for the information contained in DNA to reach the ribosomes (the site of protein synthesis) in the cell's cytoplasm, this information must first exit the nucleus. This is the purpose of messenger RNA (mRNA). In the same way a manager might dictate a letter to a secretary, transcription is the process by which the DNA code is dictated to another molecule (mRNA) that will then deliver its message into the cytoplasm.
Although there are only four nitrogenous bases, they can be arranged in countless different DNA sequences. It's this infinite variety of sequences that gives rise to the enormous diversity of life. All organisms contain the same complementary base pairs, but they are arranged in different ways.
One specific sequence of bases is termed a promoter region. To begin transcription, an enzyme (RNA polymerase) binds to a promoter region and begins to open up the DNA double helix, detaching base pairs and allowing complementary base re-pairing between one DNA strand and RNA nucleotides. RNA polymerase also connects mRNA nucleotides to form a single strand of mRNA. Recall that mRNA contains the base uracil in place of thymine. When a gene (a section of DNA) is transcribed, mRNA is created. mRNA will then direct the production of a certain protein. Each gene therefore codes for one specific protein.
Transcription of mRNA from DNA. One DNA strand serves as the template for mRNA production; the other DNA strand does not participate and is termed inactive at this transcription site. The inactive strand may, however, be actively transcribing mRNA elsewhere along its length.
Note that although only mRNA is discussed here, all three types of RNA (transfer or tRNA, and ribosomal or rRNA are the other types) are formed by transcription.
Go to the Biology 12 Web site Lesson 2.2A Protein Synthesis, Part 1: Translation to view an animation of this process. This animation will help you visualize transcription. Stop the animation when the tRNA discussion begins. You can revisit the site after the next lesson and watch the rest of the protein synthesis process.
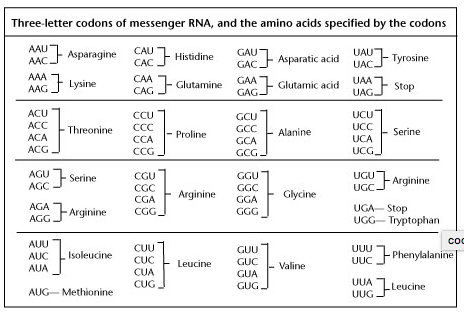
Since the newly transcribed mRNA strand will eventually code for the production of a protein, the sequence of bases in mRNA is extremely important. Every three bases along the mRNA strand will provide the code for one amino acid. These three-base groupings are triplet codons. For example, the bases A-A-G will code for the amino acid lysine. In total there are 64 possible different three-base combinations (codons), but they only code for 20 amino acids.
DNA is housed in the nucleus of a cell. In order for the information contained in DNA to reach the ribosomes (the site of protein synthesis) in the cell's cytoplasm, this information must first exit the nucleus. This is the purpose of messenger RNA (mRNA). In the same way a manager might dictate a letter to a secretary, transcription is the process by which the DNA code is dictated to another molecule (mRNA) that will then deliver its message into the cytoplasm.
Although there are only four nitrogenous bases, they can be arranged in countless different DNA sequences. It's this infinite variety of sequences that gives rise to the enormous diversity of life. All organisms contain the same complementary base pairs, but they are arranged in different ways.
One specific sequence of bases is termed a promoter region. To begin transcription, an enzyme (RNA polymerase) binds to a promoter region and begins to open up the DNA double helix, detaching base pairs and allowing complementary base re-pairing between one DNA strand and RNA nucleotides. RNA polymerase also connects mRNA nucleotides to form a single strand of mRNA. Recall that mRNA contains the base uracil in place of thymine. When a gene (a section of DNA) is transcribed, mRNA is created. mRNA will then direct the production of a certain protein. Each gene therefore codes for one specific protein.
Transcription of mRNA from DNA. One DNA strand serves as the template for mRNA production; the other DNA strand does not participate and is termed inactive at this transcription site. The inactive strand may, however, be actively transcribing mRNA elsewhere along its length.
Note that although only mRNA is discussed here, all three types of RNA (transfer or tRNA, and ribosomal or rRNA are the other types) are formed by transcription.
Go to the Biology 12 Web site Lesson 2.2A Protein Synthesis, Part 1: Translation to view an animation of this process. This animation will help you visualize transcription. Stop the animation when the tRNA discussion begins. You can revisit the site after the next lesson and watch the rest of the protein synthesis process.
Since the newly transcribed mRNA strand will eventually code for the production of a protein, the sequence of bases in mRNA is extremely important. Every three bases along the mRNA strand will provide the code for one amino acid. These three-base groupings are triplet codons. For example, the bases A-A-G will code for the amino acid lysine. In total there are 64 possible different three-base combinations (codons), but they only code for 20 amino acids.
Codons and the Amino Acids they specify
Notice that most amino acids have more than one codon associated with them, but some have six! Why have more than one codon to code for an amino acid? The answer may be that this protects the protein being formed. Any mutation that causes the last base in a codon to alter will still produce the same amino acid, and therefore still produce the correct protein.
Process of Transcription Animation
Notice that most amino acids have more than one codon associated with them, but some have six! Why have more than one codon to code for an amino acid? The answer may be that this protects the protein being formed. Any mutation that causes the last base in a codon to alter will still produce the same amino acid, and therefore still produce the correct protein.
Process of Transcription Animation
Part 2: Translation
You have learned that DNA provides the raw genetic material that will eventually code for a protein, and how DNA produces the three types of RNA (mRNA, tRNA and rRNA) required to carry out its instructions. You also know that during transcription, the DNA double helix partially unravels with the help of an enzyme, enabling the formation of a single strand of messenger RNA or mRNA. Messenger RNA, though it differs in structure from the DNA that created it, still contains the information needed to carry out protein synthesis.
Recall the numerous different types of proteins, including those that provide structural (hair, nails, and muscle), immune, endocrine, and enzyme functions in an organism. These proteins are essential for life. Messenger RNA has an important task to perform—it carries DNA's instructions from the nucleus out into the cytoplasm.
Now you will explore what happens after a mature strand of mRNA leaves the nucleus.
Translation occurs in the cytoplasm, where the purpose of DNA is fulfilled by newly created proteins that perform functions vital to the lives of cells and organisms.
After mRNA is transcribed in the nucleus, it moves into the cytoplasm to direct the formation of a polypeptide—a chain of amino acids joined by peptide bonds. A protein may be made from one or more polypeptides. While messenger RNA codes for a particular sequence of building blocks called amino acids, it also specifies the order in which they must be joined. Only 20 different amino acids exist, but they can be joined in an enormous number of different ways to create many different proteins.
So, translation is the “process by which the sequence of codons in mRNA dictates the sequence of amino acids in a polypeptide”. The players in this complex, choreographed production are mRNA, a variety of enzymes, and the two types of RNA we have yet to discuss in detail—transfer RNA (tRNA) and ribosomal RNA (rRNA).
You have learned that DNA provides the raw genetic material that will eventually code for a protein, and how DNA produces the three types of RNA (mRNA, tRNA and rRNA) required to carry out its instructions. You also know that during transcription, the DNA double helix partially unravels with the help of an enzyme, enabling the formation of a single strand of messenger RNA or mRNA. Messenger RNA, though it differs in structure from the DNA that created it, still contains the information needed to carry out protein synthesis.
Recall the numerous different types of proteins, including those that provide structural (hair, nails, and muscle), immune, endocrine, and enzyme functions in an organism. These proteins are essential for life. Messenger RNA has an important task to perform—it carries DNA's instructions from the nucleus out into the cytoplasm.
Now you will explore what happens after a mature strand of mRNA leaves the nucleus.
Translation occurs in the cytoplasm, where the purpose of DNA is fulfilled by newly created proteins that perform functions vital to the lives of cells and organisms.
After mRNA is transcribed in the nucleus, it moves into the cytoplasm to direct the formation of a polypeptide—a chain of amino acids joined by peptide bonds. A protein may be made from one or more polypeptides. While messenger RNA codes for a particular sequence of building blocks called amino acids, it also specifies the order in which they must be joined. Only 20 different amino acids exist, but they can be joined in an enormous number of different ways to create many different proteins.
So, translation is the “process by which the sequence of codons in mRNA dictates the sequence of amino acids in a polypeptide”. The players in this complex, choreographed production are mRNA, a variety of enzymes, and the two types of RNA we have yet to discuss in detail—transfer RNA (tRNA) and ribosomal RNA (rRNA).
mRNA
Messenger RNA or mRNA is a long, single strand composed of a large number of nucleotides. Recall that a nucleotide is the building block of both DNA and RNA. It contains a sugar, phosphate group, and one of four nitrogenous or nitrogen-containing bases:
DNA: adenine (A), thymine (T), cytosine (C), guanine (G)
mRNA: uracil (U) replaces thymine (T)
A codon is a three-nucleotide unit on the mRNA strand. Different codons specify different amino acids. For example, in the following chart below AAU and AAC both code for the amino acid asparagine.
DNA: adenine (A), thymine (T), cytosine (C), guanine (G)
mRNA: uracil (U) replaces thymine (T)
A codon is a three-nucleotide unit on the mRNA strand. Different codons specify different amino acids. For example, in the following chart below AAU and AAC both code for the amino acid asparagine.
tRNA
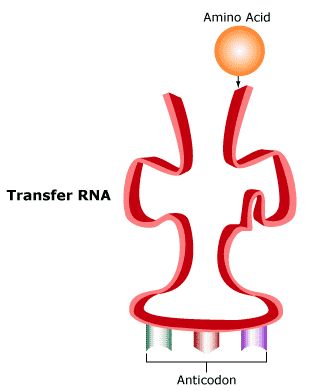
Protein synthesis occurs at the ribosomes located on rough endoplasmic reticulum. After messenger RNA leaves the nucleus, it will proceed to the ribosomes, but the amino acids that will be joined must also be present. Getting amino acids to the ribosomes is the job of transfer RNA. Transfer RNA (tRNA) is a long, single-stranded nucleic acid molecule. In tRNA, complementary base pairing gives the molecule a convoluted, boot-like shape:
The three bases in the tRNA molecule act as an anticodon that is complementary to a specific codon of mRNA. Each tRNA molecule also carries one specific amino acid to a ribosome, where protein synthesis is underway.
At the ribosomes, each tRNA anticodon binds to a specific mRNA codon. For example, the tRNA anticodon GCU would bind to the mRNA codon CGA because these bases are complementary. Remember that in RNA, A always pairs with U, and C with G.
There are three types of RNA, and two of those (mRNA and tRNA) have already been discussed. The third type is ribosomal RNA or rRNA. It is produced in the nucleolus, inside the nucleus, and is a component of ribosomal subunits that come together when protein synthesis begins.
As you will see, ribosomes move along a strand of mRNA. Often, several ribosomes will be translating the same mRNA strand at the same time, but in different locations. They move along like an assembly line. This is called a polyribosome, or polysome. Ribosomes exist only until a complete polypeptide chain is formed, then they dissociate into their subunits and fall off the mRNA strand.
Translation Animation
Translation 3 STEPS:
Step I: of protein synthesis is chain initiation, when synthesis begins. On mRNA, the codon AUG codes for methionine, the start codon. The ribosome assembles at the AUG codon along with the first tRNA molecule and its anticodon. Note that the ribosome, not the mRNA strand, will move during synthesis. Protein synthesis does not begin until a ribosome reaches the start codon (AUG) on the mRNA strand.
Step 2: chain elongation, is exactly that. A polypeptide chain grows longer as amino acids are added to it, one at a time. When a new tRNA molecule arrives at the ribosome carrying a second amino acid, it lands on the second of two binding sites on the ribosome. Methionine, the start codon, is still situated on the first binding site.
Next, methionine is peptide-bonded to the amino acid that landed with the new tRNA.
As the ribosome moves along the mRNA strand, one codon at a time (this ribosome movement is called translocation), the tRNA molecule that has delivered its amino acids leaves the ribosome. It will return again and again, each time with another amino acid to be joined to the chain. Over and over, tRNA delivers amino acids that form peptide bonds with the growing polypeptide chain.
Step 3: is chain termination. There are three codons that do not specify an amino acid. Instead, these signal stop. When the ribosome reaches a stop codon, an enzyme, also called a release factor, binds to one of the two binding sites and sets the polypeptide chain free from the last tRNA molecule.
After the production is over, the cast (ribosome, tRNA, mRNA) separates. An mRNA strand can be translated again, but it will only last as long as the polypeptide it codes for is needed.
Protein Synthesis Review Animation
Protein Synthesis Online Activity
At the ribosomes, each tRNA anticodon binds to a specific mRNA codon. For example, the tRNA anticodon GCU would bind to the mRNA codon CGA because these bases are complementary. Remember that in RNA, A always pairs with U, and C with G.
There are three types of RNA, and two of those (mRNA and tRNA) have already been discussed. The third type is ribosomal RNA or rRNA. It is produced in the nucleolus, inside the nucleus, and is a component of ribosomal subunits that come together when protein synthesis begins.
As you will see, ribosomes move along a strand of mRNA. Often, several ribosomes will be translating the same mRNA strand at the same time, but in different locations. They move along like an assembly line. This is called a polyribosome, or polysome. Ribosomes exist only until a complete polypeptide chain is formed, then they dissociate into their subunits and fall off the mRNA strand.
Translation Animation
Translation 3 STEPS:
Step I: of protein synthesis is chain initiation, when synthesis begins. On mRNA, the codon AUG codes for methionine, the start codon. The ribosome assembles at the AUG codon along with the first tRNA molecule and its anticodon. Note that the ribosome, not the mRNA strand, will move during synthesis. Protein synthesis does not begin until a ribosome reaches the start codon (AUG) on the mRNA strand.
Step 2: chain elongation, is exactly that. A polypeptide chain grows longer as amino acids are added to it, one at a time. When a new tRNA molecule arrives at the ribosome carrying a second amino acid, it lands on the second of two binding sites on the ribosome. Methionine, the start codon, is still situated on the first binding site.
Next, methionine is peptide-bonded to the amino acid that landed with the new tRNA.
As the ribosome moves along the mRNA strand, one codon at a time (this ribosome movement is called translocation), the tRNA molecule that has delivered its amino acids leaves the ribosome. It will return again and again, each time with another amino acid to be joined to the chain. Over and over, tRNA delivers amino acids that form peptide bonds with the growing polypeptide chain.
Step 3: is chain termination. There are three codons that do not specify an amino acid. Instead, these signal stop. When the ribosome reaches a stop codon, an enzyme, also called a release factor, binds to one of the two binding sites and sets the polypeptide chain free from the last tRNA molecule.
After the production is over, the cast (ribosome, tRNA, mRNA) separates. An mRNA strand can be translated again, but it will only last as long as the polypeptide it codes for is needed.
Protein Synthesis Review Animation
Protein Synthesis Online Activity
Video - Inner Workings of the Cell
ENZYMES & METABOLISM
When you think of all the activities an organism must constantly perform in order to stay alive, you might think of eating, breathing, blood circulation, excretion, and movement. However, it's also important to think about what is happening at the cellular level. Cells must continuously metabolize to produce the energy that keeps an organism alive. Depending on the type of cell, other activities might include synthesis (producing bone, muscle, and other types of tissue), breakdown (e.g., digestion), communication (e.g., neurons, hormones, etc.), and so on. These diverse biochemical reactions have something in common. They must occur at a very fast rate, and they require enzymes to maintain that rate. In short, life as we know it could not exist without enzymes!
Enzymes are proteins that are created by protein synthesis (transcription and translation), which you have already studied. Although the precise number of different proteins coded for by DNA is unknown, estimates range up to 150,000. If we accept the estimate that half of these proteins are enzymes, then there are up to 75,000 different enzymes in the human body.
Biochemical reactions are chemical reactions that occur in living organisms. These reactions begin with one or more reactants, and then convert these reactants to products. The many chemical reactions that occur in cells are collectively termed metabolism.
A typical chemical reaction might be described as:
A + B = C + D
where A and B are reactants, and C and D are products.
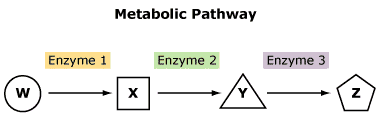
However, reactions in cells are often more complex and occur as part of a metabolic pathway—a series of conversions in which the product of one reaction becomes the reactant in the next reaction:
A g B g C g D g E g F
Note that B, C, D, and E are both reactants and products. For example, in the reaction A g B, A is the reactant and B is the product. Next, in B g C, B becomes a reactant and C is a product, and so on.
Cellular metabolism must occur rapidly in order to sustain the life of an organism. This is why metabolic pathways, such as the one above, require enzymes. Enzymes are proteins that act as catalysts. They speed up chemical reactions without being used up in those reactions. The molecules that react with enzymes are called substrates (substrate is another word for reactant). Enzymes are specific—each enzyme will only form a complex with a certain substrate. That is why different enzymes are used in different steps of a metabolic pathway.
Enzymes are proteins that are created by protein synthesis (transcription and translation), which you have already studied. Although the precise number of different proteins coded for by DNA is unknown, estimates range up to 150,000. If we accept the estimate that half of these proteins are enzymes, then there are up to 75,000 different enzymes in the human body.
Biochemical reactions are chemical reactions that occur in living organisms. These reactions begin with one or more reactants, and then convert these reactants to products. The many chemical reactions that occur in cells are collectively termed metabolism.
A typical chemical reaction might be described as:
A + B = C + D
where A and B are reactants, and C and D are products.
However, reactions in cells are often more complex and occur as part of a metabolic pathway—a series of conversions in which the product of one reaction becomes the reactant in the next reaction:
A g B g C g D g E g F
Note that B, C, D, and E are both reactants and products. For example, in the reaction A g B, A is the reactant and B is the product. Next, in B g C, B becomes a reactant and C is a product, and so on.
Cellular metabolism must occur rapidly in order to sustain the life of an organism. This is why metabolic pathways, such as the one above, require enzymes. Enzymes are proteins that act as catalysts. They speed up chemical reactions without being used up in those reactions. The molecules that react with enzymes are called substrates (substrate is another word for reactant). Enzymes are specific—each enzyme will only form a complex with a certain substrate. That is why different enzymes are used in different steps of a metabolic pathway.
Activation Energy
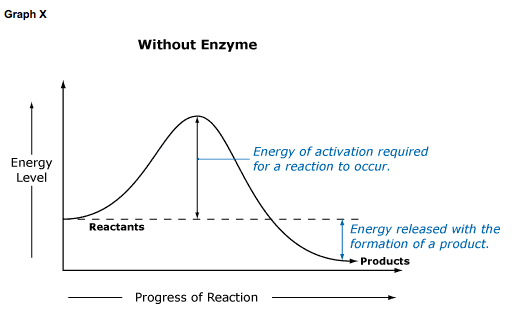
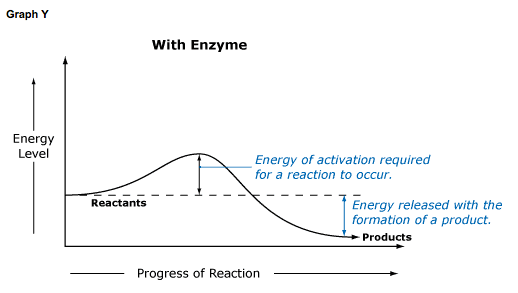
Chemical reactions cannot occur unless molecules have enough energy to react with each other. This required energy is termed energy of activation, or simply activation energy. Enzymes lower the energy of activation in a reaction, allowing it to proceed at a lower than usual energy level.
Chemical reactions cannot occur unless molecules have enough energy to react with each other. This required energy is termed energy of activation, or simply activation energy. Enzymes lower the energy of activation in a reaction, allowing it to proceed at a lower than usual energy level.
In Graph X, no enzyme is present, and the conversion of substrate to product requires a higher Ea (energy of activation) before it can occur. Graph Y shows the same reaction when an enzyme is present. Note that Ea is much smaller when an enzyme facilitates a reaction.
It is also important to note that the product of a reaction is the same whether an enzyme is present or not. However, depending on reaction conditions, more products may result, or the rate of the reaction may be altered. This will be the focus of a future lesson.
It is also important to note that the product of a reaction is the same whether an enzyme is present or not. However, depending on reaction conditions, more products may result, or the rate of the reaction may be altered. This will be the focus of a future lesson.
How Enzymes Work
Enzymes are catalysts for thousands of different biochemical reactions in the human body. Because each enzyme is specific to a certain substrate, there are many different enzymes. Although each enzyme is involved in a different reaction, all enzymes function in the same way.
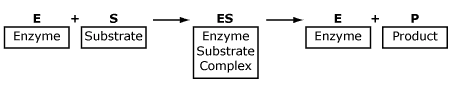
Each enzyme contains an active site to which a particular substrate can bond to form an enzyme-substrate complex, an intermediate step between reactants and products.
Each enzyme contains an active site to which a particular substrate can bond to form an enzyme-substrate complex, an intermediate step between reactants and products.
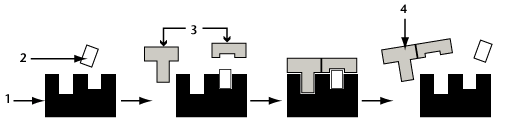
This equation is demonstrated in the following diagram.
The structure labeled 1 is an enzyme, 2 is a cofactor (more on these shortly!). Label 3 represents substrate, and 4 is the product formed by the reaction.
Note that the enzyme remains intact and is not used up. After the reaction is complete, the enzyme is available to react again. For this reason, small amounts of enzyme are often all that is required, and these small amounts can have large effects.
Refer to your Inquiry Into Life textbook and study the diagram on enzymatic action. Note the following important points:
Note that the enzyme remains intact and is not used up. After the reaction is complete, the enzyme is available to react again. For this reason, small amounts of enzyme are often all that is required, and these small amounts can have large effects.
Refer to your Inquiry Into Life textbook and study the diagram on enzymatic action. Note the following important points:
- formation of a temporary enzyme-substrate complex during reactions
- enzymes catalyze both synthesis and decomposition reactions
- enzymes are not changed or used up in reactions
|
|
Induced Fit Model
Enzyme and substrate fit together in much the same way as a lock and key, except that the enzyme actually changes itself slightly in order to fit with its substrate. Because of this slight alteration, we use the term induced fit model to better describe how the enzyme and substrate interact.
Go to your Inquiry Into Life textbook and look for an illustration of the induced fit model.
Go to the Biology 12 Web site Lesson 2.3A Enzyme Metabolism and watch a narrated animation: Navigate to Cells—Metabolism. Pay attention to the way the enzyme changes shape to induce a better fit with its substrate.
The active site on an enzyme is restored to its original shape after a product is released. Because enzymes are not used up in the reactions they catalyze, cells generally only require a small amount of a given enzyme.
Enzyme names often end in -ase and are named for their particular substrate (e.g., lipase acts upon lipids, nuclease upon nucleic acids, lactase on the sugar lactose, etc.). This does not always hold true, however. The digestive enzymes trypsin and pepsin do not follow this naming convention.
Enzyme and substrate fit together in much the same way as a lock and key, except that the enzyme actually changes itself slightly in order to fit with its substrate. Because of this slight alteration, we use the term induced fit model to better describe how the enzyme and substrate interact.
Go to your Inquiry Into Life textbook and look for an illustration of the induced fit model.
Go to the Biology 12 Web site Lesson 2.3A Enzyme Metabolism and watch a narrated animation: Navigate to Cells—Metabolism. Pay attention to the way the enzyme changes shape to induce a better fit with its substrate.
The active site on an enzyme is restored to its original shape after a product is released. Because enzymes are not used up in the reactions they catalyze, cells generally only require a small amount of a given enzyme.
Enzyme names often end in -ase and are named for their particular substrate (e.g., lipase acts upon lipids, nuclease upon nucleic acids, lactase on the sugar lactose, etc.). This does not always hold true, however. The digestive enzymes trypsin and pepsin do not follow this naming convention.
COPY CODE SNIPPET
Factors Affecting Enzyme Action
There are at least 2000 enzymes in the human body and each one is involved in a different biochemical reaction. Enzymes are essential to life because biochemical reactions cannot occur without them, so it is important to understand some of the factors that affect enzyme activity reaction rates.
In this lesson you will look at the effect on reaction rates of substrate and enzyme concentration, temperature, pH, inhibitors, and cofactors. Each of these factors can be introduced, removed, or altered to influence the rate of an enzymatic reaction.
Recall that reaction rate is the speed at which substrate is converted to product, and that biochemical reactions occur along metabolic pathways. These pathways are comprised of a specific sequence of steps that, in total, are more energy-efficient than one large reaction. Each reaction requires a specific enzyme. The more ideal the conditions for an enzyme, the more effectively it can catalyze the reaction to which it is so loyal, and the more quickly the entire pathway can proceed towards a final product.
ENZYME QUIZ
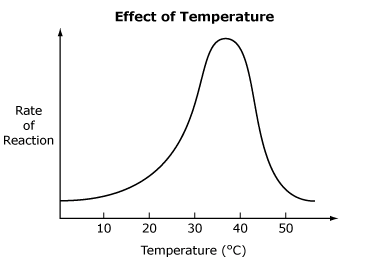
1) Temperature
Each enzyme functions most effectively at its own optimum temperature. At this optimum temperature, the movement of molecules is rapid enough to allow the maximum number of collisions between enzyme and substrate. More collisions mean an increased rate of formation of enzyme-substrate complexes. If the temperature rises too high, the enzyme becomes denatured and the reaction rate decreases. Denaturation is damage that alters the active site of an enzyme so its substrate can no longer bind to it.
There are at least 2000 enzymes in the human body and each one is involved in a different biochemical reaction. Enzymes are essential to life because biochemical reactions cannot occur without them, so it is important to understand some of the factors that affect enzyme activity reaction rates.
In this lesson you will look at the effect on reaction rates of substrate and enzyme concentration, temperature, pH, inhibitors, and cofactors. Each of these factors can be introduced, removed, or altered to influence the rate of an enzymatic reaction.
Recall that reaction rate is the speed at which substrate is converted to product, and that biochemical reactions occur along metabolic pathways. These pathways are comprised of a specific sequence of steps that, in total, are more energy-efficient than one large reaction. Each reaction requires a specific enzyme. The more ideal the conditions for an enzyme, the more effectively it can catalyze the reaction to which it is so loyal, and the more quickly the entire pathway can proceed towards a final product.
ENZYME QUIZ
1) Temperature
Each enzyme functions most effectively at its own optimum temperature. At this optimum temperature, the movement of molecules is rapid enough to allow the maximum number of collisions between enzyme and substrate. More collisions mean an increased rate of formation of enzyme-substrate complexes. If the temperature rises too high, the enzyme becomes denatured and the reaction rate decreases. Denaturation is damage that alters the active site of an enzyme so its substrate can no longer bind to it.
2) pH
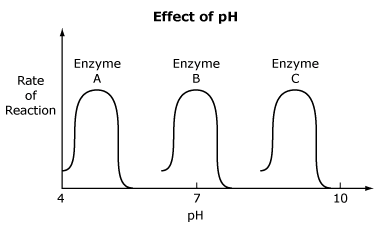
Enzymes also function best at an optimum pH. As with temperature, optimum enzyme pH varies with enzyme type and location. Recall that pH refers to the scale that indicates the acidity or alkalinity of a solution. Some enzymes, such as those involved in digestion in the stomach, function best in an acidic environment. Enzymes in the blood require a more neutral pH, and a basic pH is necessary to the activity of enzymes in the small intestine. This graph shows pH vs. the rate of reaction for three enzymes. The peaks of each curve represent the optimum pH for each enzyme. If pH is too low or too high for an enzyme, it will denature.
Enzymes also function best at an optimum pH. As with temperature, optimum enzyme pH varies with enzyme type and location. Recall that pH refers to the scale that indicates the acidity or alkalinity of a solution. Some enzymes, such as those involved in digestion in the stomach, function best in an acidic environment. Enzymes in the blood require a more neutral pH, and a basic pH is necessary to the activity of enzymes in the small intestine. This graph shows pH vs. the rate of reaction for three enzymes. The peaks of each curve represent the optimum pH for each enzyme. If pH is too low or too high for an enzyme, it will denature.
3) Enzyme & Substrate Concentration
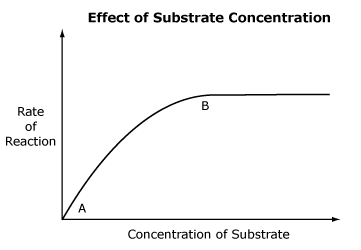
As substrate concentration increases, the rate of a reaction will also increase because substrate molecules take up more active sites. Once all available active sites are occupied, the reaction proceeds at its maximum rate (saturation point) and the graph levels off. Increased substrate concentration will have no effect on reaction rate.
As substrate concentration increases, the rate of a reaction will also increase because substrate molecules take up more active sites. Once all available active sites are occupied, the reaction proceeds at its maximum rate (saturation point) and the graph levels off. Increased substrate concentration will have no effect on reaction rate.
Between points A and B on this graph, substrate concentration increases. The rate of reaction also increases to point B. B represents active site saturation—all the enzyme available is being used in the reaction, so the reaction cannot proceed any faster. If more enzyme is added, the rate of reaction would again increase.
Increasing enzyme concentration will also increase reaction rate. More active sites are available for bonding with substrate molecules, so the chance of collisions between enzyme and substrate is higher. The reaction rate will continue to increase as long as there is enough substrate available to form enzyme-substrate complexes.
Increasing enzyme concentration will also increase reaction rate. More active sites are available for bonding with substrate molecules, so the chance of collisions between enzyme and substrate is higher. The reaction rate will continue to increase as long as there is enough substrate available to form enzyme-substrate complexes.
Enzyme Inhibition
Enzyme inhibition occurs when the substrate is unable to bind to the active site of an enzyme. The enzyme is then prevented from catalyzing biochemical reactions in the body. Depending on the enzyme, the affects of inhibition will vary. The two main types of enzyme inhibition are competitive and non-competitive.
A competitive inhibitor (a molecule similar in shape to the actual substrate) binds to the enzyme active site. The inhibitor and the substrate have to compete for the active site. The substrate molecules cannot bind with the active site if the inhibitor has already done so, and vice-versa. Some examples of competitive inhibition include:
Examples of non-competitive inhibitors include:
Enzyme inhibition occurs when the substrate is unable to bind to the active site of an enzyme. The enzyme is then prevented from catalyzing biochemical reactions in the body. Depending on the enzyme, the affects of inhibition will vary. The two main types of enzyme inhibition are competitive and non-competitive.
A competitive inhibitor (a molecule similar in shape to the actual substrate) binds to the enzyme active site. The inhibitor and the substrate have to compete for the active site. The substrate molecules cannot bind with the active site if the inhibitor has already done so, and vice-versa. Some examples of competitive inhibition include:
- In metabolic pathways, the pathway end product can bind with the first enzyme in the pathway. Since the products of one step in such a pathway become the reactants in the next step, this binding effectively blocks the pathway from proceeding. End product inhibition of metabolic pathways is almost always competitive.
- Some antibiotics work by occupying the active sites of enzymes. For example, an antibiotic interrupts the metabolic pathway that produces folic acid by competing for the active sites of one enzyme in the pathway.
- Many toxic substances act as inhibitors to enzymes involved in vital biochemical processes. Once the enzyme is inhibited the process cannot take place, and the results can include paralysis, coma, or death.
Examples of non-competitive inhibitors include:
- Heavy metal ions (lead, mercury, silver, chromium, etc.) can affect the shape of enzymes, causing heavy metal poisoning (e.g., lead poisoning from lead-based paint).
- Nerve gases such as Sarin inhibit the active site of acetylcholinesterase, an enzyme vital to proper nerve response and functioning. Nerve gas poisoning can cause death.
COPY CODE SNIPPET
Enzyme Cofactors
Just as enzymes can be inhibited, they can be helped by one of two cofactors:
Organic, non-protein coenzymes assist the enzyme in its function. Coenzymes provide atoms, ions, or electrons to enzymatic reactions. Vitamins are common components of coenzymes, so that when we lack a vitamin in our diet, coenzymes (and therefore enzymes) may not function correctly. As a result, important chemical reactions in the body may be hindered.
Just as enzymes can be inhibited, they can be helped by one of two cofactors:
- an inorganic ion (one that does not contain carbon)
- an organic, non-protein molecule called a coenzyme
Organic, non-protein coenzymes assist the enzyme in its function. Coenzymes provide atoms, ions, or electrons to enzymatic reactions. Vitamins are common components of coenzymes, so that when we lack a vitamin in our diet, coenzymes (and therefore enzymes) may not function correctly. As a result, important chemical reactions in the body may be hindered.
Unit 2 Review Questions
Please answer the following questions
1. Compare and contrast the structure and function of DNA and RNA.
2. What are the end products of:
Replication:
Transcription:
Translation:
3. Compare and contrast the function of DNA helicase with RNA polymerase.
4. What are the three steps in translation and give a brief description of each step.
5. What is the function of DNA ligase?
6. Why is the process of replication called semi-conservative?
7. During transcription, which 2 steps does RNA polymerase function?
8. At any one time, how much of the DNA is transcribed?
9. Give a description of the 3 steps involved in translation.
10. Give the sequence of base pairs below, what is the resulting amino acid sequence?
3'-TACGGCTTG-5'
11. How do enzymes increase the rates of chemical reactions?
12. An enzyme in your stomach, pepsidase, breaks peptide bonds. Its optimum pH is 2. Describe the change in reaction rate if you were to swallow a liquid which raised the pH of your stomach to 6.
13. The metabolic pathway which converts cellulose to glucose invloves 5 steps. How many different enzymes does this metabolic pathway require?
14. Explain the difference between a competitive and an non-competitive inhibitor.
15. Explain the induced fit model of enzyme activity
1. Compare and contrast the structure and function of DNA and RNA.
2. What are the end products of:
Replication:
Transcription:
Translation:
3. Compare and contrast the function of DNA helicase with RNA polymerase.
4. What are the three steps in translation and give a brief description of each step.
5. What is the function of DNA ligase?
6. Why is the process of replication called semi-conservative?
7. During transcription, which 2 steps does RNA polymerase function?
8. At any one time, how much of the DNA is transcribed?
9. Give a description of the 3 steps involved in translation.
10. Give the sequence of base pairs below, what is the resulting amino acid sequence?
3'-TACGGCTTG-5'
11. How do enzymes increase the rates of chemical reactions?
12. An enzyme in your stomach, pepsidase, breaks peptide bonds. Its optimum pH is 2. Describe the change in reaction rate if you were to swallow a liquid which raised the pH of your stomach to 6.
13. The metabolic pathway which converts cellulose to glucose invloves 5 steps. How many different enzymes does this metabolic pathway require?
14. Explain the difference between a competitive and an non-competitive inhibitor.
15. Explain the induced fit model of enzyme activity